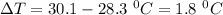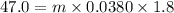Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
The specific heat of lead is 0.0380 cal/g·°C. If 47.0 calories of energy raised the temperature of a...
Questions


Mathematics, 25.05.2021 08:50

Arts, 25.05.2021 08:50

Mathematics, 25.05.2021 08:50

Mathematics, 25.05.2021 08:50

Mathematics, 25.05.2021 08:50

Mathematics, 25.05.2021 08:50



Mathematics, 25.05.2021 08:50

English, 25.05.2021 08:50




English, 25.05.2021 08:50

Mathematics, 25.05.2021 08:50

Mathematics, 25.05.2021 08:50



English, 25.05.2021 08:50

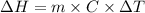
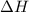 is the enthalpy change
is the enthalpy change
 is the temperature change
is the temperature change