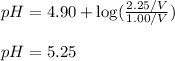Chemistry, 29.01.2020 03:45 angiechill
Abuffer solution is composed of 1.00 mol of acid and 2.25 mol of the conjugate base. if the p k a of the acid is 4.90 , what is the ph of the buffer?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 23.06.2019 01:00
Iron (fe) reacts with copper sulfate (cuso4) to form iron (ii) sulfate. in this reaction, cu2+ gains electrons to form cu. which statement is true about this reaction? fe(s) + cuso4(aq) → feso4(aq) + cu(s)
Answers: 3

Chemistry, 23.06.2019 15:30
How many grams of c3h8 is needed in the reactants to produce 10.5 mil of h2o
Answers: 2

Chemistry, 23.06.2019 17:30
Which of the following elements would you expect to have the highest ionization energy value, and why? a. chlorine (cl), because it has a low effective nuclear charge and large radius b. fluorine (f), because it has a large radius and naturally forms a negative ion c. lithium (li), because it has a small radius and naturally forms a positive ion d. neon (ne), because it has a high effective nuclear charge and small radius
Answers: 2
You know the right answer?
Abuffer solution is composed of 1.00 mol of acid and 2.25 mol of the conjugate base. if the p k a of...
Questions



Mathematics, 17.07.2019 14:30

Social Studies, 17.07.2019 14:30


Mathematics, 17.07.2019 14:30

Mathematics, 17.07.2019 14:30

History, 17.07.2019 14:30

Mathematics, 17.07.2019 14:30


Mathematics, 17.07.2019 14:30

History, 17.07.2019 14:30

Chemistry, 17.07.2019 14:30

Mathematics, 17.07.2019 14:30


Biology, 17.07.2019 14:30

History, 17.07.2019 14:30

History, 17.07.2019 14:30

Social Studies, 17.07.2019 14:30

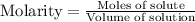
![pH=pK_a+\log(\frac{[\text{conjugate base}]}{[acid]})](/tpl/images/0480/0680/2cb39.png)
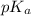 = negative logarithm of acid dissociation constant of weak acid = 4.90
= negative logarithm of acid dissociation constant of weak acid = 4.90![[\text{conjugate base}]=\frac{2.25}{V}](/tpl/images/0480/0680/63641.png)
![[acid]=\frac{1.00}{V}](/tpl/images/0480/0680/42281.png)