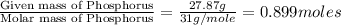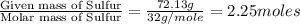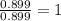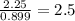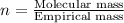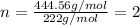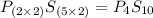Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Which of the following best explains why the end of a spoon sticking out of a cup of hot water also gets hot? question 7 options: the heat from the hot water is conducted through the spoon handle the hot water heats the air surrounding the upper part of the spoon. the hot water causes a physical change in the spoon handle. the hot water causes a chemical reaction to take place in the spoon.
Answers: 2

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Ferrophosphorus (fe2p) reacts with pyrite (fes2), producing iron (ii) sulfide and a compound that is...
Questions


History, 22.12.2019 02:31


History, 22.12.2019 02:31

Mathematics, 22.12.2019 02:31






Mathematics, 22.12.2019 02:31

Mathematics, 22.12.2019 02:31

Mathematics, 22.12.2019 02:31

Biology, 22.12.2019 02:31

Mathematics, 22.12.2019 02:31





Mathematics, 22.12.2019 02:31

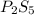 and
and 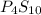 respectively.
respectively.