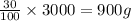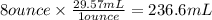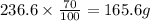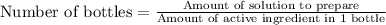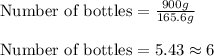Chemistry, 23.01.2020 21:31 kaykay9243
Apharmacy intern is asked to prepare 3 l of a 30% w/v solution. t he pharmacy stocks the active ingredient in 8-ounce bottles of 70% w/v strength. how many bottles will be needed as the source of the active ingredient?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2


Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
You know the right answer?
Apharmacy intern is asked to prepare 3 l of a 30% w/v solution. t he pharmacy stocks the active ingr...
Questions


English, 23.10.2019 17:10

History, 23.10.2019 17:10


Advanced Placement (AP), 23.10.2019 17:10


Social Studies, 23.10.2019 17:10



Social Studies, 23.10.2019 17:10

History, 23.10.2019 17:10

History, 23.10.2019 17:10

History, 23.10.2019 17:10

Biology, 23.10.2019 17:10


Mathematics, 23.10.2019 17:10

History, 23.10.2019 17:10

Mathematics, 23.10.2019 17:10

History, 23.10.2019 17:10

Geography, 23.10.2019 17:10