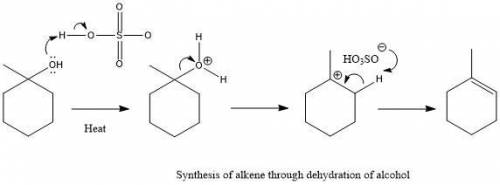
Chemistry, 23.01.2020 07:31 allieallie
In the presence of sulfuric acid, this alcohol is dehydrated to form an alkene through an e1 mechanism. in the box, draw the major alkene product of this reaction.

Answers: 2


Another question on Chemistry

Chemistry, 20.06.2019 18:02
1.milk of magnesia, a suspension of mg(oh)2in water, reacts with stomach acid (hcl) in a neutralization reaction. mg(oh)2(s) + 2 hcl(aq) −→ 2 h2o(l) + mgcl2(aq) what mass of mgcl2 will be produced if 5.49 g of mg(oh)2 reacts? answer in units of g. 2.what mass of hcl is required to completely react with 5.49 g of mg(oh)2? answer in units of g.
Answers: 3


Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3
You know the right answer?
In the presence of sulfuric acid, this alcohol is dehydrated to form an alkene through an e1 mechani...
Questions

Physics, 23.03.2020 21:27

Mathematics, 23.03.2020 21:27



Chemistry, 23.03.2020 21:27


Computers and Technology, 23.03.2020 21:27



Computers and Technology, 23.03.2020 21:27



English, 23.03.2020 21:27

Mathematics, 23.03.2020 21:27


Social Studies, 23.03.2020 21:27

Mathematics, 23.03.2020 21:27

History, 23.03.2020 21:27


Mathematics, 23.03.2020 21:27