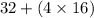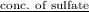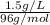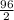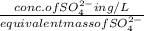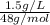Chemistry, 16.01.2020 18:31 jacksoneaton
An analytical chemist determines that an estuarine water sample contains 1.5 g/liter of sulfate ion (so4 2). what is the concentration in terms of: (a) g/l of sulfur? (b) molar concentration of sulfate? (c) normality?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which sentence best describes the formation of igneous rock? a- lava on the surface dries up and makes arock b_melted rocks cools and forms crystals c_rocks under tremendous heat and pressure d_magma is melted rock underground
Answers: 1

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3
You know the right answer?
An analytical chemist determines that an estuarine water sample contains 1.5 g/liter of sulfate ion...
Questions


History, 13.03.2021 01:00






Social Studies, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00

Computers and Technology, 13.03.2021 01:00

Social Studies, 13.03.2021 01:00

Mathematics, 13.03.2021 01:00


Mathematics, 13.03.2021 01:00

Biology, 13.03.2021 01:00




Chemistry, 13.03.2021 01:00

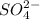 is as follows.
is as follows.