
Chemistry, 14.01.2020 19:31 kelseydavid69
How many grams of iron can be obtained from a 268-g sample of iron(iii) oxide?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 17:30
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1

You know the right answer?
How many grams of iron can be obtained from a 268-g sample of iron(iii) oxide?...
Questions


Social Studies, 24.09.2019 00:00


Mathematics, 24.09.2019 00:00


Health, 24.09.2019 00:00


Biology, 24.09.2019 00:00

English, 24.09.2019 00:00




Chemistry, 24.09.2019 00:00



Mathematics, 24.09.2019 00:00




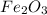 as:-
as:-
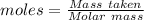
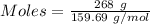
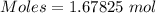
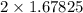 moles of iron.
moles of iron. 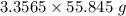 = 187.44 g
= 187.44 g

