rate = k[n_2]^3 [o_3]

Chemistry, 14.01.2020 00:31 helphelp81
The rate of a certain reaction is given by the following rate law:
rate = k[n_2]^3 [o_3]
use this information to answer the questions below.
1. what is the reaction order in n_2?
2. what is the reaction order in o_3?
3. what is overall reaction order?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2

Chemistry, 22.06.2019 22:00
Does the number of ions in solution increase, decrease, or remain constant? it continuously decreases. it continuously increases. it decreases at first, then increases. it increases at first, then decreases.
Answers: 3
You know the right answer?
The rate of a certain reaction is given by the following rate law:
rate = k[n_2]^3 [o_3]
rate = k[n_2]^3 [o_3]
Questions

Chemistry, 16.11.2020 22:00

World Languages, 16.11.2020 22:00

Chemistry, 16.11.2020 22:00

Business, 16.11.2020 22:00

History, 16.11.2020 22:00

Advanced Placement (AP), 16.11.2020 22:00

Chemistry, 16.11.2020 22:00





Computers and Technology, 16.11.2020 22:00

Mathematics, 16.11.2020 22:00

Mathematics, 16.11.2020 22:00

Mathematics, 16.11.2020 22:00


Arts, 16.11.2020 22:00

Mathematics, 16.11.2020 22:00


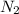 = 3
= 3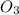 = 1
= 1![rate = k[N_2]^3 [O_3]](/tpl/images/0453/5258/fc831.png)


