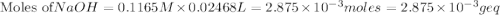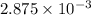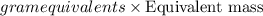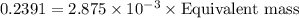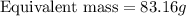Chemistry, 09.01.2020 06:31 roseemariehunter12
If is found that 24.68 ml of .1165 m naoh is needed to titrate .2931 g of an unknown acid to the phenolphthalein end point. calculate the equivlanet mass of the acid.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 23.06.2019 09:00
A2-kg bowling ball is 1 meter off the ground on a post when it falls. just before it reaches the ground,its traveling 4.4 m/s. assuming that there is no air resistant, which statement is true a. the initial potential energy is less then the final kinetic energy b. the mechanical energy is not conserved c. the mechanical energy is conserved d. the initial potential energy is greater than the final kinetic energy
Answers: 3
You know the right answer?
If is found that 24.68 ml of .1165 m naoh is needed to titrate .2931 g of an unknown acid to the phe...
Questions


Law, 07.05.2020 05:57


English, 07.05.2020 05:57




Mathematics, 07.05.2020 05:57


Mathematics, 07.05.2020 05:57



Arts, 07.05.2020 05:57

Mathematics, 07.05.2020 05:57



Mathematics, 07.05.2020 05:57



English, 07.05.2020 05:57

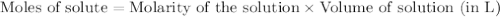
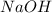 solution = 0.1165 M
solution = 0.1165 M