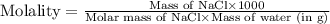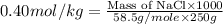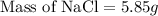Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

You know the right answer?
How many grams of nacl should be added to 250 grams of waterto
make a 0.40 m solutions?...
make a 0.40 m solutions?...
Questions

Mathematics, 05.05.2021 23:50


Chemistry, 05.05.2021 23:50

Social Studies, 05.05.2021 23:50


Mathematics, 05.05.2021 23:50






Spanish, 05.05.2021 23:50

Physics, 05.05.2021 23:50

History, 05.05.2021 23:50




Social Studies, 05.05.2021 23:50

Health, 05.05.2021 23:50

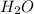 = 250 g
= 250 g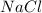 = 58.5 g/mole
= 58.5 g/mole