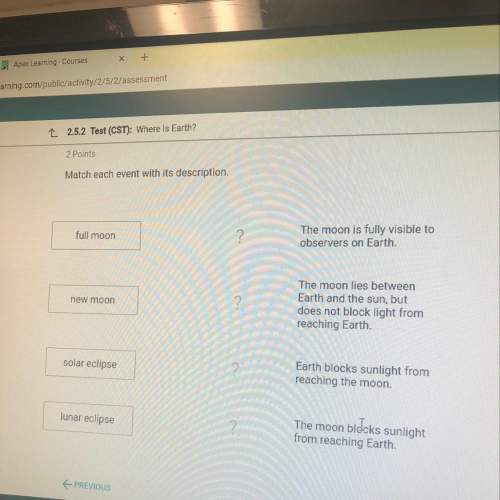
2.5.2 test (cst): where is earth?
question 6 of 15
2 points
match each event wit...


Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
This image is an example of a(n) a) atom. b) compound. c) mixture. d) molecule.
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 19:30
Describe the forces both attractive and repulsive that occur as two atoms move closer together.
Answers: 1
You know the right answer?
Questions

Mathematics, 09.12.2020 04:00


Geography, 09.12.2020 04:00



History, 09.12.2020 04:00

Arts, 09.12.2020 04:00


Chemistry, 09.12.2020 04:00



Mathematics, 09.12.2020 04:00

History, 09.12.2020 04:00

Social Studies, 09.12.2020 04:00

Mathematics, 09.12.2020 04:00

Chemistry, 09.12.2020 04:00

Law, 09.12.2020 04:00

Mathematics, 09.12.2020 04:00


Mathematics, 09.12.2020 04:00