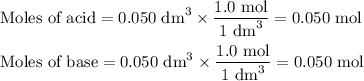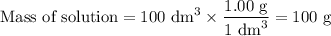Chemistry, 05.01.2020 17:31 gharrell03
50cm3 of 1 mol/dm3 hcl at 30°c was mixed with 50cm3 of 1mol/dm3 naoh at 30°c in a styrofoam calorimeter. the temperature of the calorimeter rose by 4.5°c. calculate the heat of reaction per mol of h20 formed.( heat capacity of the calorimeter is 50j/°c

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1

Chemistry, 23.06.2019 05:00
1. true or false: minerals are inorganic. true false 2. inorganic means that something has never been found alive 3. halite is another name for and is a mineral with a cubic crystal pattern. table salt rock salt
Answers: 2
You know the right answer?
50cm3 of 1 mol/dm3 hcl at 30°c was mixed with 50cm3 of 1mol/dm3 naoh at 30°c in a styrofoam calorime...
Questions

English, 21.04.2021 19:00







Chemistry, 21.04.2021 19:00


Mathematics, 21.04.2021 19:00

Mathematics, 21.04.2021 19:00

Mathematics, 21.04.2021 19:00


Mathematics, 21.04.2021 19:00


Mathematics, 21.04.2021 19:00


Mathematics, 21.04.2021 19:00

Social Studies, 21.04.2021 19:00