
Assuming the metals lose all their valence electrons and the nonmetals gain electrons to complete the s-p subshells, which listing below shows the correct information for the element? elements electron structure of atom electron structure of ion net ion charge: 1. sodium 1s22s22p63s1 1s22s22p6 -12. fluorine 1s22s22p4 1s22s22p6 -13. aluminum 1s22s22p63s23p1 1s22s22p6 +3a. the listing for sodium is correct. b. the listing for fluorine is correct. c. the listing for aluminum is correct. d. none of these

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
In numbering carbon atoms in the parent chain of a hydrocarbon, why would you number from right to left, rather than left to right
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2


You know the right answer?
Assuming the metals lose all their valence electrons and the nonmetals gain electrons to complete th...
Questions

Business, 28.07.2019 14:30


History, 28.07.2019 14:30

Social Studies, 28.07.2019 14:30

History, 28.07.2019 14:30




Biology, 28.07.2019 14:30

Biology, 28.07.2019 14:30

Biology, 28.07.2019 14:30

Social Studies, 28.07.2019 14:30







Social Studies, 28.07.2019 14:30

History, 28.07.2019 14:30

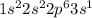 . When sodium loses one electron then it will attain +1 charge and its electronic configuration will be as follows.
. When sodium loses one electron then it will attain +1 charge and its electronic configuration will be as follows.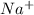 :
: 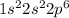
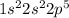 . When fluorine gains an electron then it acquires -1 charge and its electronic configuration is as follows.
. When fluorine gains an electron then it acquires -1 charge and its electronic configuration is as follows.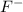 :
: 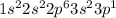 . When aluminium loses its valence electrons then it acquires +3 charge and its electronic configuration is as follows.
. When aluminium loses its valence electrons then it acquires +3 charge and its electronic configuration is as follows.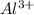 :
: 

