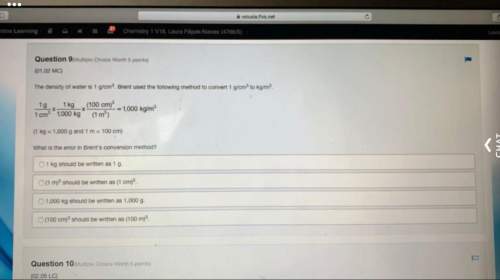
Phosphorus tribromide decomposes to form phosphorus and bromide, like this:
4pbr3(>...

Chemistry, 28.12.2019 01:31 coontcakes
Phosphorus tribromide decomposes to form phosphorus and bromide, like this:
4pbr3(> p4(g)+6br2(g)
also, a chemist finds that a certain temperature at the equilibrium mixture of phosphorus tribromide, phosphorus, and bromine has the following composition:
compound pressure at equiibrium
pbr3 97.4 atm
p4 99.2 atm
br2 97.2 atm
calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 05:20
Identify and describe the three ways that mutations affect organisms.
Answers: 1

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1
You know the right answer?
Questions







History, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01



Biology, 05.10.2020 15:01


Advanced Placement (AP), 05.10.2020 15:01

Mathematics, 05.10.2020 15:01


History, 05.10.2020 15:01



Chemistry, 05.10.2020 15:01