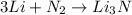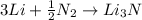Lithium metal reacts with nitrogen gas to form lithium nitride. identify the balanced reaction that describes this process. a. li + n lin b. li + n_2 lin_2 c. 2li + n_2 li_2n_2 d. 6li + n_2 2li_2n e. 2li + n_2 2lin 64. which of the elements listed below is most likely to exhibit an expanded octet in its compounds? a. o, p b. s, p c. na, k d. c, s e. n, s

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Which of the following statements is true? question 4 options: nuclear decay rates vary with the conditions of the reaction, but chemical reaction rates do not. chemical reaction rates vary with the conditions of the reaction, but nuclear decay rates do not. neither chemical reaction rates nor nuclear decay rates vary with the conditions of the reaction. both chemical reaction rates and nuclear decay rates vary with the conditions of the reaction.
Answers: 1



Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2
You know the right answer?
Lithium metal reacts with nitrogen gas to form lithium nitride. identify the balanced reaction that...
Questions

Mathematics, 04.06.2021 18:30






Mathematics, 04.06.2021 18:30


Mathematics, 04.06.2021 18:30

Mathematics, 04.06.2021 18:30

Mathematics, 04.06.2021 18:30


Mathematics, 04.06.2021 18:30


Mathematics, 04.06.2021 18:30


Mathematics, 04.06.2021 18:30


History, 04.06.2021 18:30

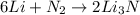
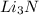 . It consists of
. It consists of 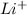 and
and 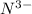 ions.
ions.