
pleaeee
which of the following explains the vsepr geometry of a carbon tetrachloride molecule? (5 points)
it is bent because there are four bonded pairs around carbon.
it is tetrahedral because there are four bonded pairs around carbon.
it is bent because there are two bonded pairs and two lone pairs around carbon.
it is tetrahedral because there are two bonded pairs and two lone pairs around carbon.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1

Chemistry, 22.06.2019 17:00
The msds for glacial acetic acid says that it is a flammable liquid that can severely burn any human tissue it comes in contact with. it reacts with bases, various metals, and strong oxidizing agents. its vapors can form explosive mixtures with air.
Answers: 1

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2
You know the right answer?
pleaeee
which of the following explains the vsepr geometry of a carbon tetrachloride mol...
which of the following explains the vsepr geometry of a carbon tetrachloride mol...
Questions

Mathematics, 20.09.2020 02:01





History, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01


Mathematics, 20.09.2020 02:01

English, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01


Mathematics, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01


Chemistry, 20.09.2020 02:01

English, 20.09.2020 02:01

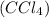 has carbon atom as central atom surrounded by four chlorine atoms.
has carbon atom as central atom surrounded by four chlorine atoms.

