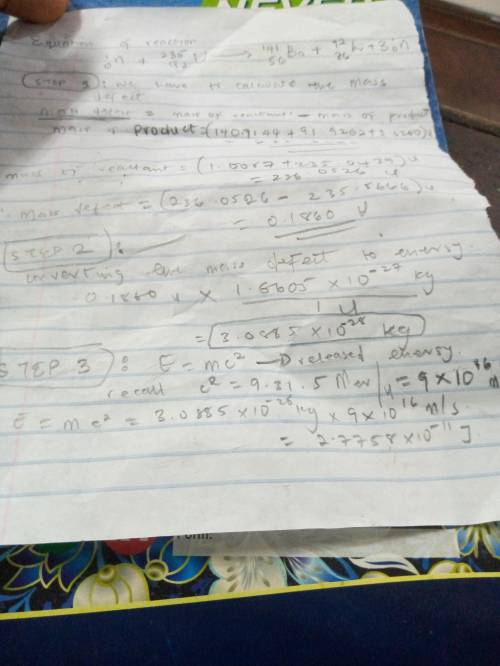Chemistry, 20.12.2019 22:31 mannyrocks1532
What is the reaction energy q of this reaction? use c2=931.5mev/u. express your answer in millions of electron volts to three significant figures. view available hint(s) qq q = nothing mev part b using fission, what mass m of uranium-235 would be necessary to supply all of the energy that the united states uses in a year, roughly 1.0×1019j? express your answer in kilograms to two significant figures.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Janel’s class studied properties of matter and how matter can change. janel decided she would do an experiment mixing baking soda and vinegar.question: describe the properties of baking soda and vinegar, and explain the changes that janel should see when she mixes the two types of matter. •first, identify the physical state of matter of baking soda. describe another property of baking soda. •next, identify the physical state of matter of vinegar. describe another property of vinegar. •then, explain what janel should see when she mixes the baking soda and vinegar. •describe the states of matter of the new materials that are formed. •explain how janel can be certain a change has occurred. me
Answers: 3

Chemistry, 22.06.2019 12:00
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
What is the reaction energy q of this reaction? use c2=931.5mev/u. express your answer in millions...
Questions

Social Studies, 19.01.2021 01:00




Mathematics, 19.01.2021 01:00

History, 19.01.2021 01:00


Mathematics, 19.01.2021 01:00


Computers and Technology, 19.01.2021 01:00

English, 19.01.2021 01:00


Mathematics, 19.01.2021 01:00



Mathematics, 19.01.2021 01:00

Arts, 19.01.2021 01:00



English, 19.01.2021 01:00