
Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 03:00
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1
You know the right answer?
Ineed on both a and b of question 1
...
...
Questions

Mathematics, 24.01.2020 20:31

Mathematics, 24.01.2020 20:31

History, 24.01.2020 20:31

Computers and Technology, 24.01.2020 20:31



Mathematics, 24.01.2020 20:31


Physics, 24.01.2020 20:31

English, 24.01.2020 20:31

English, 24.01.2020 20:31

History, 24.01.2020 20:31


Mathematics, 24.01.2020 20:31

Computers and Technology, 24.01.2020 20:31

Mathematics, 24.01.2020 20:31


English, 24.01.2020 20:31


Biology, 24.01.2020 20:31


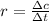
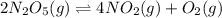
![r = -\frac{\Delta [N_2O_5]}{2 \Delta t}](/tpl/images/0427/5608/fed8d.png)
![r = \frac{\Delta [NO_2]}{4 \Delta t} = \frac{\Delta [O_2]}{\Delta t}](/tpl/images/0427/5608/b318c.png)
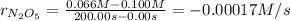
![-\frac{\Delta [N_2O_5]}{2 \Delta t} = \frac{\Delta [NO_2]}{4 \Delta t}](/tpl/images/0427/5608/d754b.png)
![r_{NO_2} = \frac{\Delta [NO_2]}{\Delta t}](/tpl/images/0427/5608/bc64c.png)
![-\frac{4 \Delta [N_2O_5]}{2 \Delta t} = \frac{\Delta [NO_2]}{\Delta t}](/tpl/images/0427/5608/7f9a6.png)


