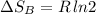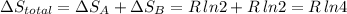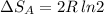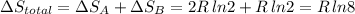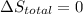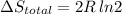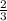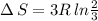Chemistry, 20.12.2019 04:31 blakesmith0110
Arigid container is divided into two compartments of equal volume by a partition. one compartment contains 1 mole of ideal gas a at 1 atm, and the other contains 1 mole of ideal gas b at 1 atm. calculate the increase in entropy which occurs when the partition between the two compartments is removed. if the frst compartment had contained 2 moles of ideal gas a, what would have been the increase in entropy when the partition was removed? calculate the corresponding increases in entropy in each of the preceding two situations if both compartments had contained ideal gas a.

Answers: 3


Another question on Chemistry


Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1
You know the right answer?
Arigid container is divided into two compartments of equal volume by a partition. one compartment co...
Questions








English, 18.03.2020 02:56


History, 18.03.2020 02:56


English, 18.03.2020 02:56




Mathematics, 18.03.2020 02:57




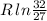 .
.