Chemistry, 17.12.2019 04:31 princessx7543
Given that  [br(g)] = 111.9 kj ⋅ mol⁻¹
[br(g)] = 111.9 kj ⋅ mol⁻¹ [br(g)] = 111.9 kj⋅mol⁻¹
[br(g)] = 111.9 kj⋅mol⁻¹ [c(g)] = 716.7 kj ⋅ mol⁻¹
[c(g)] = 716.7 kj ⋅ mol⁻¹ [c(g)] = 716.7 kj⋅mol⁻¹
[c(g)] = 716.7 kj⋅mol⁻¹ [cbr₄(g)] = 29.4 kj ⋅ mol⁻¹
[cbr₄(g)] = 29.4 kj ⋅ mol⁻¹ [cbr₄(g)] = 29.4 kj⋅mol⁻¹ calculate the average molar bond enthalpy of the carbon–bromine bond in a cbr₄ molecule.
[cbr₄(g)] = 29.4 kj⋅mol⁻¹ calculate the average molar bond enthalpy of the carbon–bromine bond in a cbr₄ molecule.

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1
You know the right answer?
Given that [tex]\delta h^o_f[/tex] [br(g)] = 111.9 kj ⋅ mol⁻¹[tex]\delta h^o_f[/tex] [br(g)] = 111.9...
Questions

History, 21.05.2020 05:08




Computers and Technology, 21.05.2020 05:08

Chemistry, 21.05.2020 05:08

Mathematics, 21.05.2020 05:08

Mathematics, 21.05.2020 05:08


Spanish, 21.05.2020 05:08


Mathematics, 21.05.2020 05:08






History, 21.05.2020 05:08


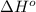
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f(product)]-\sum [n\times \Delta H^o_f(reactant)]](/tpl/images/0421/8523/45485.png)
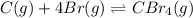
![\Delta H^o_{rxn}=[(n_{(CBr_4)}\times \Delta H^o_f_{(CBr_4)})]-[(n_{(Br)}\times \Delta H^o_f_{(Br)})+(n_{(C)}\times \Delta H^o_f_{(C)})]](/tpl/images/0421/8523/304b7.png)
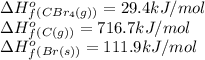
![\Delta H^o_{rxn}=[(1\times 29.4)]-[(4\times 111.9)+(1\times 716.7)]=-1134.9kJ/mol](/tpl/images/0421/8523/33ec8.png)
![\Delta H=-[4\times B.E_{C-Br}]](/tpl/images/0421/8523/b980a.png)
![-1134.9=-[4\times B.E_{C-Br}]](/tpl/images/0421/8523/74d8f.png)