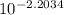Chemistry, 13.12.2019 03:31 smithsa10630
Electrochemical cells can be used to measure ionic concentrations. a cell is set up with a pair of zn electrodes, each immersed in znso4 solution. one solution is created to be exactly 0.500 m, and the potential on its electrode is measured to be 0.065 v higher than the other electrode. what is the concentration of the other solution? enter your answer in m.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 22.06.2019 19:50
What is the wavelength of a wave with a velocity of 50 m/s and a frequency of 5hz a 250 m b 0.1 m c 10m d 0.01 m
Answers: 2

Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3

Chemistry, 23.06.2019 01:10
Volume is a measurement of how fast particles of a substance are moving
Answers: 3
You know the right answer?
Electrochemical cells can be used to measure ionic concentrations. a cell is set up with a pair of z...
Questions

History, 19.02.2020 05:46

Mathematics, 19.02.2020 05:46




Mathematics, 19.02.2020 05:46

Geography, 19.02.2020 05:46






Mathematics, 19.02.2020 05:46


Mathematics, 19.02.2020 05:47





Mathematics, 19.02.2020 05:47