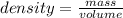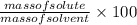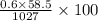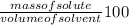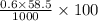Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 23.06.2019 01:00
Which statement best describes isomers? a. isomers are alcohols that have the same functional group. b. isomers have at least one carbon-carbon double bond. c. isomers have the same molecular formula but different structural properties.
Answers: 1
You know the right answer?
Ocean water is about 0.600m nacl and has a density of 1.027g/ml. find the percentage of nacl (w/w) a...
Questions


Mathematics, 07.02.2021 22:10

Mathematics, 07.02.2021 22:10



Mathematics, 07.02.2021 22:10


Mathematics, 07.02.2021 22:10




Mathematics, 07.02.2021 22:10

Mathematics, 07.02.2021 22:10

Geography, 07.02.2021 22:10




Mathematics, 07.02.2021 22:10

Mathematics, 07.02.2021 22:10