
Chemistry, 11.12.2019 02:31 bethanybowers4986
Ineed asap i need to go make christmas presents after
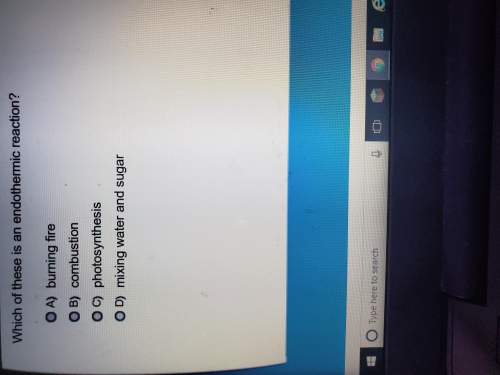
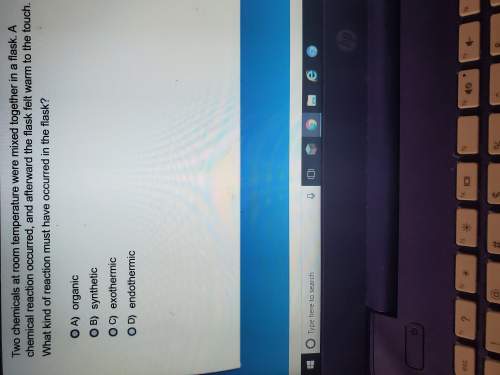
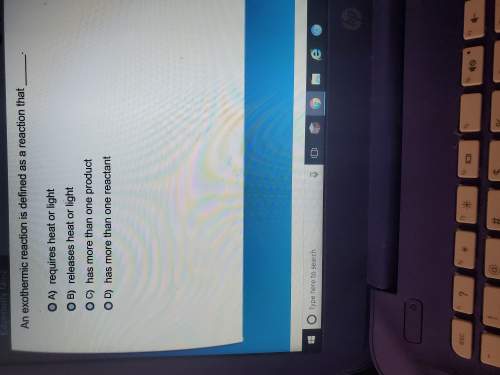

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3

Chemistry, 23.06.2019 18:30
Esure to answer all parts. the equilibrium constant for the reaction ni2+(aq) + 6 nh3(aq) ⇌ ni(nh3)6 2+(aq) is kf = 5.6 × 108 at 25°c. (a) what is δg o at this temperature? (b) if standard-state concentrations of reactants and products are mixed, in which direction does the reaction proceed? (c) determine δg when [ni(nh3)62+] = 0.010 m, [ni2+] = 0.0010 m, and [nh3] = 0.0050 m. in which direction will the reaction proceed to achieve equilibrium? (a) × 10 j/mol (enter your answer in scientific notation.) (b) to the right. to the left. (c) × 10 j/mol (enter your answer in scientific notation.) to the right. to the left.
Answers: 3


Chemistry, 24.06.2019 02:00
1. what is an isomer? how many possible isomers of hexane are there? what are the structural differences between these isomers?
Answers: 1
You know the right answer?
Ineed asap i need to go make christmas presents after
...
...
Questions




English, 24.10.2020 14:00

Chemistry, 24.10.2020 14:00

Computers and Technology, 24.10.2020 14:00

Chemistry, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00

Computers and Technology, 24.10.2020 14:00

English, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00


English, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00


English, 24.10.2020 14:00

Chemistry, 24.10.2020 14:00



