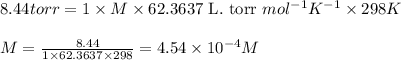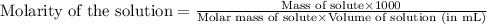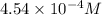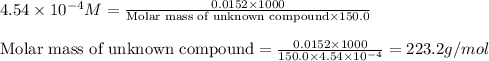Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1


Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
A150.0 ml sample of an aqueous solution at 25°c contains 15.2 mg of an unknown nonelectrolyte compou...
Questions

Arts, 19.09.2019 05:50


Social Studies, 19.09.2019 05:50

Biology, 19.09.2019 05:50

History, 19.09.2019 05:50


Business, 19.09.2019 05:50



Mathematics, 19.09.2019 05:50

History, 19.09.2019 05:50

Social Studies, 19.09.2019 05:50


Biology, 19.09.2019 05:50

Biology, 19.09.2019 06:00

Social Studies, 19.09.2019 06:00


Business, 19.09.2019 06:00

Mathematics, 19.09.2019 06:00

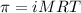
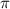 = osmotic pressure of the solution = 8.44 torr
= osmotic pressure of the solution = 8.44 torr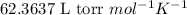
![25^oC=[273+25]=298K](/tpl/images/0411/3120/6a9f9.png)