
Chemistry, 07.12.2019 03:31 jc95704816
Balance the chemical equation given below, and determine the number of milliliters of 0.0300m phosphoric acid required to neutralize 25.00ml of 0.0150m calcium hydroxide.
__ca(oh)2(aq) + __h3po4(aq) --> __ca3(po4)2(s) + __h2o(l)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
When determining the shape of a molecule, it is important to draw a lewis dot structure first in order to see the total number a. electrons within the moleculeb. bonding and unshared pairs around central atomc. unshared pair within the molecule( i really need it )
Answers: 1

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3
You know the right answer?
Balance the chemical equation given below, and determine the number of milliliters of 0.0300m phosph...
Questions





Biology, 10.07.2019 08:00

Biology, 10.07.2019 08:00




Mathematics, 10.07.2019 08:00


Mathematics, 10.07.2019 08:00

History, 10.07.2019 08:00


Biology, 10.07.2019 08:00

Mathematics, 10.07.2019 08:00



English, 10.07.2019 08:00

Chemistry, 10.07.2019 08:00

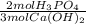 = 2,50x10⁻⁴ moles of H₃PO₄.
= 2,50x10⁻⁴ moles of H₃PO₄. 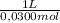 = 8,33x10⁻³L ≡ 8,33mL of 0,0300M phosphoric acid
= 8,33x10⁻³L ≡ 8,33mL of 0,0300M phosphoric acid

