
Chemistry, 03.12.2019 22:31 ronnie7898
A3.452 g sample containing an unknown amount of a ce(iv) salt is dissolved in 250.0 ml of 1 m h2so4. a 25.00 ml aliquot is analyzed by adding ki and titrating the i3^- that forms with s2o3^2-. the end point was reached following the addition of 13.02 ml of 0.03247 m na2s2o3. calculate the weight percent of ce^4+ in the sample?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 23.06.2019 04:40
Equal numbers of moles of he(g), ar(g), and ne(g) are placed in a glass vessel at room temperature. if the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixture has effused?
Answers: 1

Chemistry, 23.06.2019 10:10
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3

Chemistry, 23.06.2019 13:30
These traits describe either a chemical or a nuclear reaction. which statements describe a nuclear reaction? check all that apply. involves the loss, gain, or sharing of electrons may involve a change in total mass involve relatively low energy changes occur outside the nucleus involve very high-energy changes involve changes in nuclides when decay occurs
Answers: 1
You know the right answer?
A3.452 g sample containing an unknown amount of a ce(iv) salt is dissolved in 250.0 ml of 1 m h2so4....
Questions




Arts, 11.07.2019 04:30





Advanced Placement (AP), 11.07.2019 04:30


English, 11.07.2019 04:30





History, 11.07.2019 04:30


Social Studies, 11.07.2019 04:30


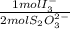 = 2,114x10⁻⁴ moles of I₃⁻
= 2,114x10⁻⁴ moles of I₃⁻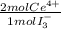 = 4,228x10⁻⁴ moles of Ce(IV).
= 4,228x10⁻⁴ moles of Ce(IV). 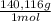 = 0,05924 g of Ce(IV)
= 0,05924 g of Ce(IV)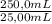 =0,5924g of Ce(IV) in the sample
=0,5924g of Ce(IV) in the sample

