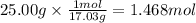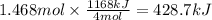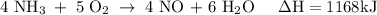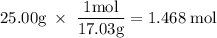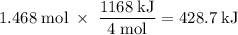Chemistry, 03.12.2019 04:31 averylivinglife2041
How much heat is absorbed/released when 25.00 g of nh3(g) reacts in the presence of excess o2(g) to produce no(g) and h2o(l) according to the following chemical equation? 4 nh3(g) + 5 o2(g) → 4 no(g) + 6 h2o(l) δh° = 1168 kj

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1

Chemistry, 23.06.2019 01:30
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3

Chemistry, 23.06.2019 08:00
Which term means two or more atoms that share electrons in a chemical bond? a. hydrogen bond b. moleculec. ionic bondd. element amd you
Answers: 3
You know the right answer?
How much heat is absorbed/released when 25.00 g of nh3(g) reacts in the presence of excess o2(g) to...
Questions



History, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10







English, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10

English, 18.03.2021 03:10

History, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10