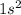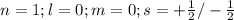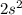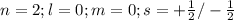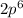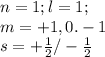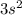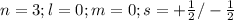Chemistry, 28.11.2019 06:31 moningersavannah
The electron configuration of a neutral atom is 1s22s22p63s2. write a complete set of quantum numbers for each of the electrons. name the element.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:30
10. according to the law of conservation of mass, how does the mass of the products in a chemical reaction compare to the mass of the reactants?
Answers: 3

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1
You know the right answer?
The electron configuration of a neutral atom is 1s22s22p63s2. write a complete set of quantum number...
Questions



Physics, 12.07.2019 08:00

English, 12.07.2019 08:00

History, 12.07.2019 08:00


Mathematics, 12.07.2019 08:00

Mathematics, 12.07.2019 08:00

Mathematics, 12.07.2019 08:00

Mathematics, 12.07.2019 08:00







Mathematics, 12.07.2019 08:00


Social Studies, 12.07.2019 08:00