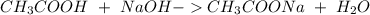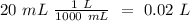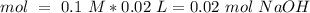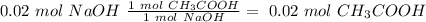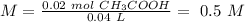Chemistry, 26.11.2019 01:31 jsjsjsskakwkowwj
40.0 ml of an acetic acid of unknown concentration is titrated with 0.100 m naoh. after 20.0 ml of the base solution has been added, the ph in the titration flask is 5.10. what was the concentration of the original acetic acid solution? (ka(ch3cooh) = 1.8 × 10–5)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1
You know the right answer?
40.0 ml of an acetic acid of unknown concentration is titrated with 0.100 m naoh. after 20.0 ml of t...
Questions

English, 01.04.2021 17:20


Mathematics, 01.04.2021 17:20

Mathematics, 01.04.2021 17:20

Mathematics, 01.04.2021 17:20


Mathematics, 01.04.2021 17:20

Mathematics, 01.04.2021 17:20

Mathematics, 01.04.2021 17:20

Chemistry, 01.04.2021 17:20


Mathematics, 01.04.2021 17:20


Mathematics, 01.04.2021 17:20

Arts, 01.04.2021 17:20

English, 01.04.2021 17:20


Mathematics, 01.04.2021 17:20

Mathematics, 01.04.2021 17:20

Mathematics, 01.04.2021 17:20