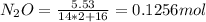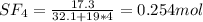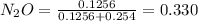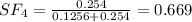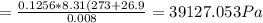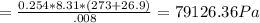A8.00 l tank at 26.9 c is filled with 5.53 g of dinitrogen difluoride gas and 17.3 g of sulfur hexafluoride gas. you can assume both gases behave as ideal gases under these conditions. calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1
You know the right answer?
A8.00 l tank at 26.9 c is filled with 5.53 g of dinitrogen difluoride gas and 17.3 g of sulfur hexaf...
Questions

Engineering, 26.10.2020 07:30

Mathematics, 26.10.2020 07:30

English, 26.10.2020 07:30


Mathematics, 26.10.2020 07:40





Mathematics, 26.10.2020 07:40






Mathematics, 26.10.2020 07:40


Chemistry, 26.10.2020 07:40