Consider the following reversible reaction.
what is the equilibrium constant expression...

Chemistry, 24.11.2019 08:31 vapelordcarl69
Consider the following reversible reaction.
what is the equilibrium constant expression for the given system?
1st pic is equation, 2nd is a, 3rd is b, 4th is c, and 5th is d.
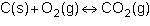
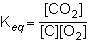
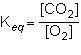
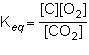
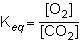

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Based on the law of conservation of energy, which statement is false? answer- energy is lost when machines dont work right
Answers: 1

Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 23.06.2019 04:50
The diagin dilutepage 6 of 12a6a5(a)fluorine, chlorine, bromine and iodine are placed in the same group of theperiodic table.state the common name used to describe elements in this group.(i)state the group in which the elements are placed and explain whythey are placed in that group.(ii)which of the above named elements is a solid at roomtemperature and pressure?
Answers: 2
You know the right answer?
Questions




Mathematics, 25.06.2019 21:30






Computers and Technology, 25.06.2019 21:30







Mathematics, 25.06.2019 21:30


English, 25.06.2019 21:30

Mathematics, 25.06.2019 21:30



