3.01 x 103 j

Chemistry, 23.11.2019 17:31 tordoor7182
The average kinetic energy of 1 mole of a gas at -32 degrees celsius is:
3.01 x 103 j
3.99 x 102 j
-3.99 x 102 j
3.80 x 103 j

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 15:30
Why does earth rotate? because earth is formed from cold gases collapsing due to gravity because the matter in the nebula that formed earth was spinning because earth forms more than 99% of the mass of the solar system because the hydrogen atoms inside the nebula fused to form helium
Answers: 1

Chemistry, 23.06.2019 00:20
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1
You know the right answer?
The average kinetic energy of 1 mole of a gas at -32 degrees celsius is:
3.01 x 103 j
3.01 x 103 j
Questions


Biology, 19.09.2020 01:01

Mathematics, 19.09.2020 01:01



Geography, 19.09.2020 01:01

Mathematics, 19.09.2020 01:01


Chemistry, 19.09.2020 01:01





Mathematics, 19.09.2020 01:01




Physics, 19.09.2020 01:01


Chemistry, 19.09.2020 01:01

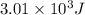
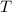 be the temperature in kelvin.
be the temperature in kelvin.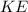 be the average kinetic energy.
be the average kinetic energy. be the number of moles.
be the number of moles.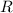 be the universal gas constant.
be the universal gas constant.
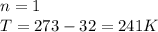
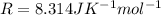
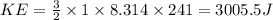 =
=

