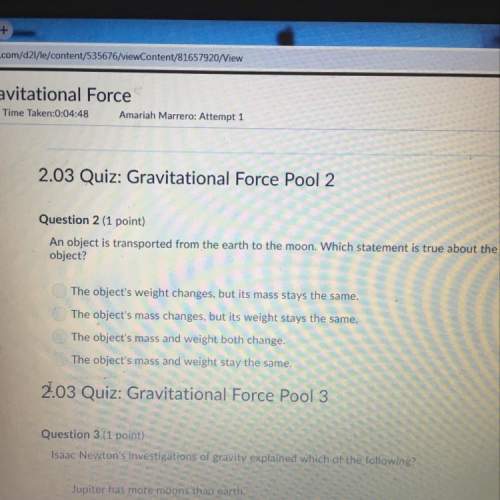
Chemistry, 22.11.2019 01:31 NeverEndingCycle
For each of the following, indicate the type of reaction (substitution; 1,1-insertion; 1,2-insertion; deinsertion; oxidative addition; reductive elimination; nucleophilic addition to a ligand; electrophilic addition to a ligand; or abstraction) and the electron count on the metal before and after the reaction. (a) os3(co)11(ch3cn) + c2h4 > os3(co)11(ƞ2-c2h4) + ch3cn
(b) (ƞ5-c5h5)rh(ch2ch3)(pme3)+ > (ƞ5-c5h5)rh(h)(pme3)+ + ch2=ch2
(c) (oc)5w=c(ome)ch3 + bbr3 > (oc)5w=cch3+ + b(ome)br3-
(d) (me3p)2cume + co2 > (me3p)2cu{o-c(=o)me}

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:10
Which is true of transition metals when moving from left to right on the periodic table? the d sublevels are not filled across the period. the cation radii become larger across the period. atomic radii increase slightly and then start to decrease. atomic radii decrease slightly and then start to increase. o
Answers: 2

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

You know the right answer?
For each of the following, indicate the type of reaction (substitution; 1,1-insertion; 1,2-inserti...
Questions

Mathematics, 27.10.2020 18:00

History, 27.10.2020 18:00




Physics, 27.10.2020 18:00

Social Studies, 27.10.2020 18:00

English, 27.10.2020 18:00

Social Studies, 27.10.2020 18:00


English, 27.10.2020 18:00







Mathematics, 27.10.2020 18:00

Mathematics, 27.10.2020 18:00