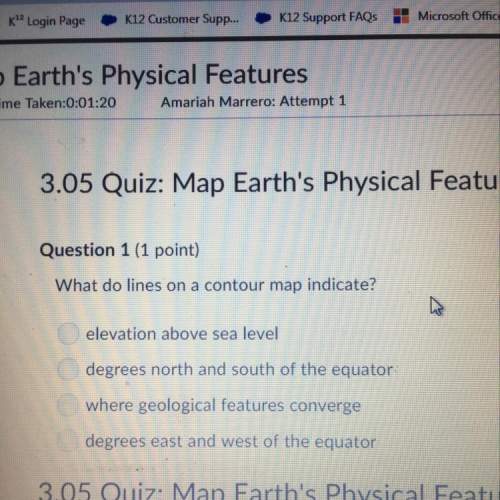
Compounds x and y are both c7h15cl products formed in the radical chlorination of 2,4-dimethylpentane. base-promoted e2 elimination of x and y gives, in each case, a single c7h14 alkene. both x and y undergo an sn2 reaction with sodium iodide in acetone solution to give c7h15i products; in this reaction y reacts faster than x. what is the structure of x?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1

Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2
You know the right answer?
Compounds x and y are both c7h15cl products formed in the radical chlorination of 2,4-dimethylpentan...
Questions

Social Studies, 03.02.2020 09:55



English, 03.02.2020 09:55

Mathematics, 03.02.2020 09:55

Mathematics, 03.02.2020 09:55

Mathematics, 03.02.2020 09:55

Spanish, 03.02.2020 09:55

Chemistry, 03.02.2020 09:55

History, 03.02.2020 09:55








English, 03.02.2020 09:55