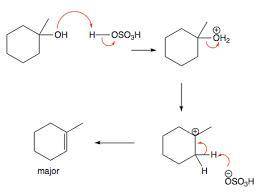
Chemistry, 14.11.2019 17:31 spreadlight10
Why was it necessary to wash the crude methylcyclohexenes with aqueous sodium bicarbonate

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2


Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
Why was it necessary to wash the crude methylcyclohexenes with aqueous sodium bicarbonate...
Questions


Mathematics, 22.03.2020 02:49



History, 22.03.2020 02:49

Mathematics, 22.03.2020 02:49







English, 22.03.2020 02:50


Computers and Technology, 22.03.2020 02:50


English, 22.03.2020 02:51



Advanced Placement (AP), 22.03.2020 02:51