
Chemistry, 13.11.2019 00:31 staz13wiggins
The following sequence of reactions occurs in the commercial production of aqueous nitric acid. (the overall industrial process produces excess no.) 4 nh3(g) + 5 o2(g) → 4 no(g) + 6 h2o(l) δh = −1166.0 kj/mol (1) 2 no(g) + o2(g) → 2 no2(g) δh = −116.2 kj/mol (2) 3 no2(g) + h2o(l) → 2 hno3(aq) + no(g) δh = −137.3 kj/mol (3) determine the total energy change (in kj) for the production of one mole of aqueous nitric acid by this process.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1
You know the right answer?
The following sequence of reactions occurs in the commercial production of aqueous nitric acid. (the...
Questions

Mathematics, 29.01.2021 16:30

Physics, 29.01.2021 16:30



Mathematics, 29.01.2021 16:30



English, 29.01.2021 16:30




Mathematics, 29.01.2021 16:30

Biology, 29.01.2021 16:30

Mathematics, 29.01.2021 16:30

History, 29.01.2021 16:30


Social Studies, 29.01.2021 16:30



Mathematics, 29.01.2021 16:30

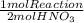 = 709,8 kJ
= 709,8 kJ

