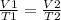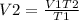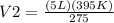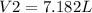Chemistry, 10.11.2019 22:31 ernestinebell3514
The temperature of a balloon rises from 275°k to 395°k, what would the ending volume be if it started at 5 l?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
The temperature of a balloon rises from 275°k to 395°k, what would the ending volume be if it starte...
Questions

Mathematics, 09.04.2021 01:00

Mathematics, 09.04.2021 01:00

Mathematics, 09.04.2021 01:00


History, 09.04.2021 01:00


Mathematics, 09.04.2021 01:00

History, 09.04.2021 01:00




History, 09.04.2021 01:00

English, 09.04.2021 01:00

Mathematics, 09.04.2021 01:00


Advanced Placement (AP), 09.04.2021 01:00

Mathematics, 09.04.2021 01:00

Mathematics, 09.04.2021 01:00


Mathematics, 09.04.2021 01:00

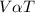 Mathematically,
Mathematically, 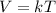 , thus,
, thus, 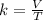 Therefore, at varying temperature and volume;
Therefore, at varying temperature and volume;