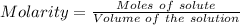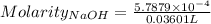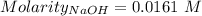Chemistry, 10.11.2019 06:31 maddietomlinson113
Ask your teacher sodium hydroxide solution is usually standardized by titrating a pure sample of potassium hydrogen phthalate (khc8h4o4, often abbreviated khp), an acid with one acidic hydrogen and a molar mass of 204.220 g/mol. it takes 36.01 ml of a sodium hydroxide solution to titrate a 0.1182-g sample of khp. what is the molarity of the sodium hydroxide

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2


Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1
You know the right answer?
Ask your teacher sodium hydroxide solution is usually standardized by titrating a pure sample of pot...
Questions

Mathematics, 17.03.2020 04:25







Social Studies, 17.03.2020 04:25






Mathematics, 17.03.2020 04:25

Mathematics, 17.03.2020 04:25

English, 17.03.2020 04:25


Computers and Technology, 17.03.2020 04:25


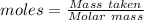
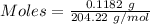
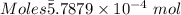
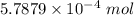 of KHP reacts with
of KHP reacts with