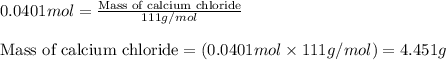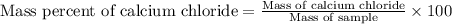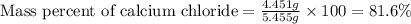Chemistry, 09.11.2019 08:31 Caixiayang3613
1. a 5.455-g sample of impure cacl2 is dissolved and treated with excess potassium carbonate solution. the dried caco3 (calcium carbonate) precipitate weighs 4.010-g. calculate the percent by mass of cacl2 in the original mixture.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:10
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 23.06.2019 05:30
The term gas is limited to those substances that exist in the gaseous state at
Answers: 1

Chemistry, 23.06.2019 06:20
An object of mass 10.0 kg and volume 1000 ml and density 10 g/ml sinks in water who’s density is 1.0 g/ml. what is the mass of the water which has been displaced in kilograms
Answers: 1
You know the right answer?
1. a 5.455-g sample of impure cacl2 is dissolved and treated with excess potassium carbonate solutio...
Questions













Social Studies, 15.12.2020 16:10



Business, 15.12.2020 16:10




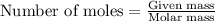 .....(1)
.....(1)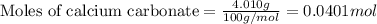
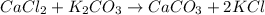
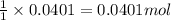 of calcium chloride
of calcium chloride