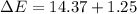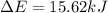Chemistry, 02.11.2019 03:31 Jsanders2276
In a piston, the addition of 14.37 kj of heat to a 100. g sample of a liquid at a constant temperature of 35.2 °c caused the liquid to vaporize (change to a gas). the vaporized gas expanded against an external pressure of 1.07 atm and a volume change of 11.49 l was observed. (recall: 1 l• atm = 101.3 j} what was the change in the internal energy of the system, (ae in kj)? (enter your answer with two decimals places and no units.)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
The speed of light is around 6.706×10^8 miles per hour. what is the speed of light in units of miles per minute?
Answers: 2

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1
You know the right answer?
In a piston, the addition of 14.37 kj of heat to a 100. g sample of a liquid at a constant temperatu...
Questions



English, 02.08.2019 07:00

Social Studies, 02.08.2019 07:00

History, 02.08.2019 07:00

Spanish, 02.08.2019 07:00


Mathematics, 02.08.2019 07:00



Mathematics, 02.08.2019 07:00

Mathematics, 02.08.2019 07:00

Chemistry, 02.08.2019 07:00

Chemistry, 02.08.2019 07:00

Mathematics, 02.08.2019 07:00


Mathematics, 02.08.2019 07:00

History, 02.08.2019 07:00

History, 02.08.2019 07:00

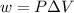
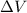 = change in volume = 11.49 L
= change in volume = 11.49 L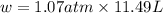
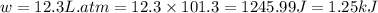 (as per conversion)
(as per conversion)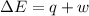
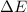 = internal energy of the system
= internal energy of the system