
Chemistry, 02.09.2019 17:00 monkemily1
Astraight-chain hydrocarbon that has only one double bond in each molecule has the general formula
(1) cnh2n-6 (3) cnh2n
(2) cnh2n-2 (4) cnh2n+2

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3
You know the right answer?
Astraight-chain hydrocarbon that has only one double bond in each molecule has the general formula
Questions

Mathematics, 29.06.2019 14:30

Biology, 29.06.2019 14:30

Physics, 29.06.2019 14:30

Biology, 29.06.2019 14:30

English, 29.06.2019 14:30

Biology, 29.06.2019 14:30

Mathematics, 29.06.2019 14:30

World Languages, 29.06.2019 14:30




Mathematics, 29.06.2019 14:30

Mathematics, 29.06.2019 14:30




Mathematics, 29.06.2019 14:30

History, 29.06.2019 14:30

Spanish, 29.06.2019 14:30

Mathematics, 29.06.2019 14:30

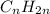
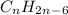 is not a valid formula for an alkane, alkene or alkyne.
is not a valid formula for an alkane, alkene or alkyne. 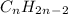 is the general formula for an alkyne that has a triple bond between carbon atoms. The example for an alkyne is acetylene and it's formula is
is the general formula for an alkyne that has a triple bond between carbon atoms. The example for an alkyne is acetylene and it's formula is 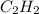 .
. 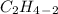
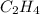
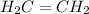
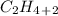
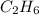
 , it has a single bond between carbons and so it is an alkane.
, it has a single bond between carbons and so it is an alkane.

