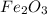Given the balanced equation representing a reaction:
fe2o3 + 2al--> al2o3 + 2fe
dur...

Chemistry, 24.01.2020 14:31 goverton101
Given the balanced equation representing a reaction:
fe2o3 + 2al--> al2o3 + 2fe
during this reaction, the oxidation number of fe changes from
(1) +2 to 0 as electrons are transferred
(2) +2 to 0 as protons are transferred
(3) +3 to 0 as electrons are transferred
(4) +3 to 0 as protons are transferred

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1


Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3
You know the right answer?
Questions







Mathematics, 24.06.2020 02:01




Mathematics, 24.06.2020 02:01


Mathematics, 24.06.2020 02:01


Mathematics, 24.06.2020 02:01

Business, 24.06.2020 02:01



Biology, 24.06.2020 02:01