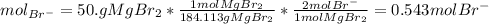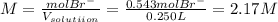Achemist prepares a solution of magnesium bromide by measuring out of into a volumetric flask and filling to the mark with distilled water. calculate the molarity of anions in the chemist's solution. be sure your answer is rounded to the correct number of significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:40
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1


Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 23.06.2019 03:30
The molar mass of iron(fe) is 55.8 g/mol. what is the mass in grams of 2.25 moles of iron?
Answers: 1
You know the right answer?
Achemist prepares a solution of magnesium bromide by measuring out of into a volumetric flask and fi...
Questions




Biology, 16.12.2020 21:30







Mathematics, 16.12.2020 21:30

Mathematics, 16.12.2020 21:30


Social Studies, 16.12.2020 21:30


Mathematics, 16.12.2020 21:30


Mathematics, 16.12.2020 21:30

Physics, 16.12.2020 21:30

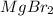 and the volume of the volumetric flask are not given, one can assume arbitrary values and you could modify them whenever you want, thus, let 50.0g of magnesium bromide to be the measured out amount of solute and a 250-mL volumetric flask where the solution is prepared, so the final volume of the solution is 250 mL after the addition of distilled water. In this manner, the bromide anions moles, taking into account there are two bromide moles per magnesium bromide moles, turns out into:
and the volume of the volumetric flask are not given, one can assume arbitrary values and you could modify them whenever you want, thus, let 50.0g of magnesium bromide to be the measured out amount of solute and a 250-mL volumetric flask where the solution is prepared, so the final volume of the solution is 250 mL after the addition of distilled water. In this manner, the bromide anions moles, taking into account there are two bromide moles per magnesium bromide moles, turns out into: