
You have a mixture of 3-fluorophenol and ethyl-4-aminobenzoate that you wish to separate by extraction. the mixture is dissolved in methylene chloride. why can you not use nahco3 to separate one component from the other? (explain why nahco3 cannot either component into the aqueous layer).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2
You know the right answer?
You have a mixture of 3-fluorophenol and ethyl-4-aminobenzoate that you wish to separate by extracti...
Questions

Mathematics, 03.07.2019 10:30



Business, 03.07.2019 10:30







Physics, 03.07.2019 10:30

English, 03.07.2019 10:30

Biology, 03.07.2019 10:30

Computers and Technology, 03.07.2019 10:30

Biology, 03.07.2019 10:30





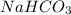 contains majorly
contains majorly 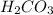 in solution.Ethyl-4-amino benzoate does not suffer side reaction e.g. ester hydrolysis with
in solution.Ethyl-4-amino benzoate does not suffer side reaction e.g. ester hydrolysis with 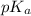 value around 8.4 and
value around 8.4 and 

