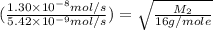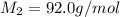Chemistry, 29.10.2019 19:31 Hippiekoolaid
Methane (ch4) effuses through a small opening in the side of a container at the rate of 1.30 × 10 8 mol s 1. an unknown gas effuses through the same opening at the rate of 5.42 × 10 9 mol s 1 when maintained at the same tem- perature and pressure as the methane. determine the molar mass of the unknown gas.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1

Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
You know the right answer?
Methane (ch4) effuses through a small opening in the side of a container at the rate of 1.30 × 10 8...
Questions




Mathematics, 26.03.2020 01:07



Mathematics, 26.03.2020 01:07


Mathematics, 26.03.2020 01:07


Mathematics, 26.03.2020 01:08



History, 26.03.2020 01:08



English, 26.03.2020 01:08

Health, 26.03.2020 01:08

Mathematics, 26.03.2020 01:08

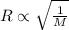
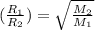 ..........(1)
..........(1)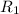 = rate of effusion of methane gas =
= rate of effusion of methane gas = 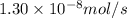
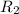 = rate of effusion of unknown gas =
= rate of effusion of unknown gas = 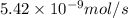
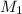 = molar mass of methane gas = 16 g/mole
= molar mass of methane gas = 16 g/mole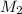 = molar mass of unknown gas = ?
= molar mass of unknown gas = ?