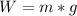Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 04:00
4. absorption has the highest risk of overdose due to increased potency. a. rectal b. oral c. transdermal d. intranasal
Answers: 2

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
You know the right answer?
What properties of matter explain why the weight of an object on the moon is different than the weig...
Questions




Mathematics, 21.10.2019 21:10

Mathematics, 21.10.2019 21:10

Mathematics, 21.10.2019 21:10





Health, 21.10.2019 21:10

Mathematics, 21.10.2019 21:10

History, 21.10.2019 21:10



Biology, 21.10.2019 21:10

Biology, 21.10.2019 21:10

History, 21.10.2019 21:10


Chemistry, 21.10.2019 21:10