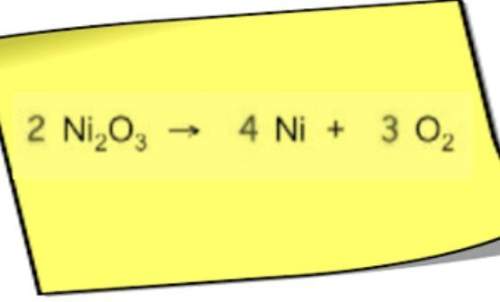
Chemistry, 24.10.2019 06:43 aletadaboss
Calculate the molarity of 139 grams of sucrose, c12h22o11, in 2.60 liters of solution.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 18:00
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3
You know the right answer?
Calculate the molarity of 139 grams of sucrose, c12h22o11, in 2.60 liters of solution....
Questions






Physics, 05.07.2019 05:30



Mathematics, 05.07.2019 05:30




Mathematics, 05.07.2019 05:30

Biology, 05.07.2019 05:30


Mathematics, 05.07.2019 05:30


Business, 05.07.2019 05:30

Business, 05.07.2019 05:30