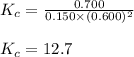At equilibrium, the concentrations of reactants and products can be predicted using the equilibrium constant, kc, which is a mathematical expression based on the chemical equation. for example, in the reaction aa+bb⇌cc+dd where a, b, c, and d are the stoichiometric coefficients, the equilibrium constant is kc=[c]c[d]d[a]a[b]b where [a], [b], [c], and [d] are the equilibrium concentrations. if the reaction is not at equilibrium, the quantity can still be calculated, but it is called the reaction quotient, qc, instead of the equilibrium constant, kc. qc=[c]tc[d]td[a]ta[b]tb where each concentration is measured at some arbitrary time t. part a a mixture initially contains a, b, and c in the following concentrations: [a] = 0.350 m , [b] = 0.800 m , and [c] = 0.500 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.150 m and [c] = 0.700 m . calculate the value of the equilibrium constant, kc. express your answer numerically.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1
You know the right answer?
At equilibrium, the concentrations of reactants and products can be predicted using the equilibrium...
Questions




Biology, 20.03.2020 22:48


Social Studies, 20.03.2020 22:48



Mathematics, 20.03.2020 22:49


Mathematics, 20.03.2020 22:49




Mathematics, 20.03.2020 22:49

Mathematics, 20.03.2020 22:49

Computers and Technology, 20.03.2020 22:50

History, 20.03.2020 22:50


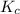
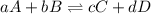
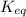 is written as:
is written as:![K_{eq}=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0304/8312/9c8b0.png)
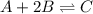
![[A]_{eq}=0.150M](/tpl/images/0304/8312/2394d.png)
![[C]_{eq}=0.700M](/tpl/images/0304/8312/9e4dd.png)
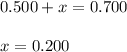
![[B]_{eq}=(0.800-x)=0.800-0.200=0.600M](/tpl/images/0304/8312/eb193.png)
![K_c=\frac{[C]}{[A][B]^2}](/tpl/images/0304/8312/240ef.png)