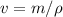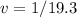Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
You know the right answer?
The density of pure gold is 19.3g/cm3. what is the volume of 1.00 g of pure gold?...
Questions

Mathematics, 06.09.2020 23:01

Chemistry, 06.09.2020 23:01


Mathematics, 06.09.2020 23:01

Mathematics, 06.09.2020 23:01



English, 06.09.2020 23:01



Physics, 06.09.2020 23:01

Mathematics, 06.09.2020 23:01

Chemistry, 06.09.2020 23:01

Mathematics, 06.09.2020 23:01


Mathematics, 06.09.2020 23:01

Biology, 06.09.2020 23:01

English, 06.09.2020 23:01

English, 06.09.2020 23:01

Mathematics, 06.09.2020 23:01

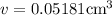
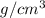
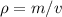
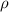 =density of pure gold
=density of pure gold