
38. a sample of calcium chloride, cacl2, has a mass of 27.8 g (ref: p.330-340)
mokes of compound
a) how many moles of calcium chloride are in this mass? show all steps and work leading to your
final answer.
b) how many chloride ions are present? show all steps and work leading to your final answer.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3


Chemistry, 23.06.2019 00:30
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1
You know the right answer?
38. a sample of calcium chloride, cacl2, has a mass of 27.8 g (ref: p.330-340)
mokes of compo...
mokes of compo...
Questions

Mathematics, 19.04.2021 19:30

Social Studies, 19.04.2021 19:30

Biology, 19.04.2021 19:30

Mathematics, 19.04.2021 19:30


Mathematics, 19.04.2021 19:30


Mathematics, 19.04.2021 19:30

Social Studies, 19.04.2021 19:30





Chemistry, 19.04.2021 19:30

Arts, 19.04.2021 19:30


History, 19.04.2021 19:30

Mathematics, 19.04.2021 19:30


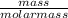 (1)
(1)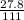 = 0.2505mole
= 0.2505mole

